In this article, we will compare this promising technology against the existing standards for diagnosing and testing Lyme disease. 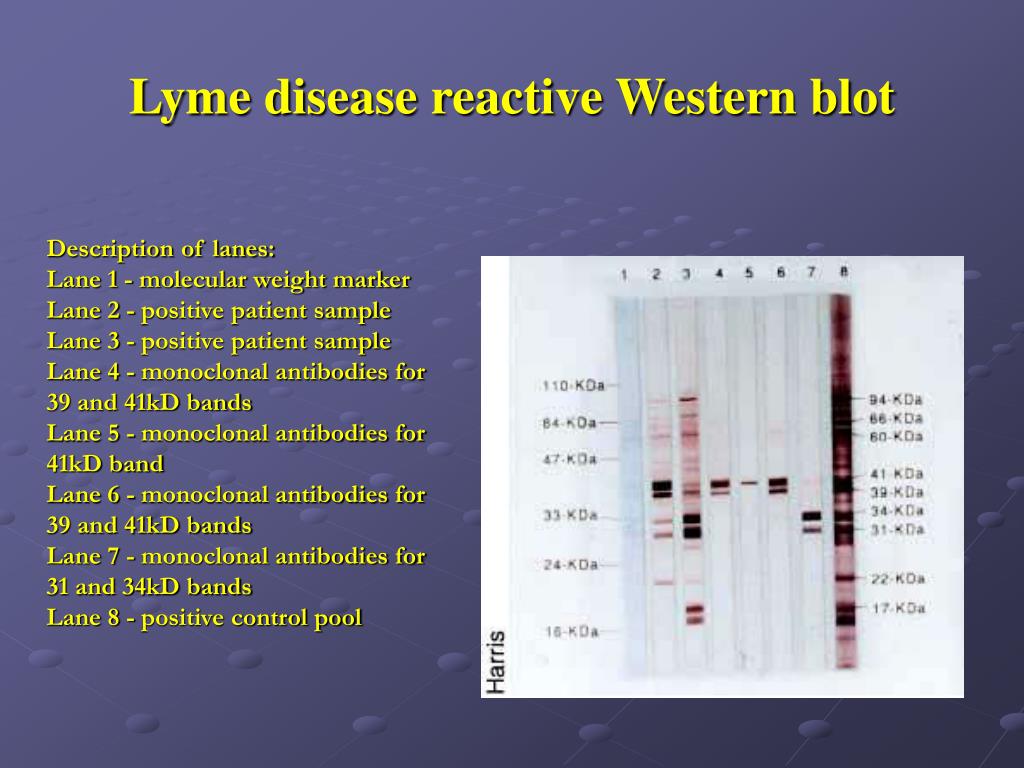
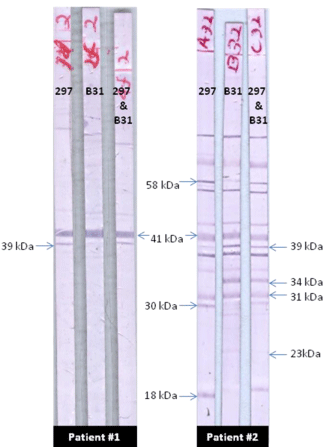
Thus, it allows for rapid results and specific identification of tick-borne illnesses. PathoDNA is a Clinical Laboratory Improvement and Amendments (CLIA)-validated laboratory test that achieves these results utilizing Next Generational DNA Sequencing and a proprietary bioinformatics database. burgdorferi and 95% or greater for other common tick-borne pathogens with superior sensitivity and selectivity. PathoDNA, a Next-Generation DNA sequencing test for Lyme disease and other tick-borne pathogens, achieves accuracy rates of 98% for B. While these practices have been a staple of microbiology for decades, these accuracy rates are unacceptable for diagnostic tests when better technology is available. The two-step test, an ELISA immunoblot followed by a confirmatory Western blot, yields accuracy rates as low as 29% for acute Lyme disease and 75% for chronic Lyme disease. This leads to increasing medical expenses and worse outcomes for patients. A general misunderstanding of Lyme disease and overconfidence in the Center for Disease Control’s (CDC) recommended two-step test for Lyme disease has led to misdiagnosis and incorrect treatment over the years. The controversial subject of chronic Lyme disease has occupied medical discourse for years while contributing to unprecedented patient suffering in the United States and abroad.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
